Discover the versatile applications of Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide, a precision tool designed for enhanced accuracy in ultrasound-guided procedures.
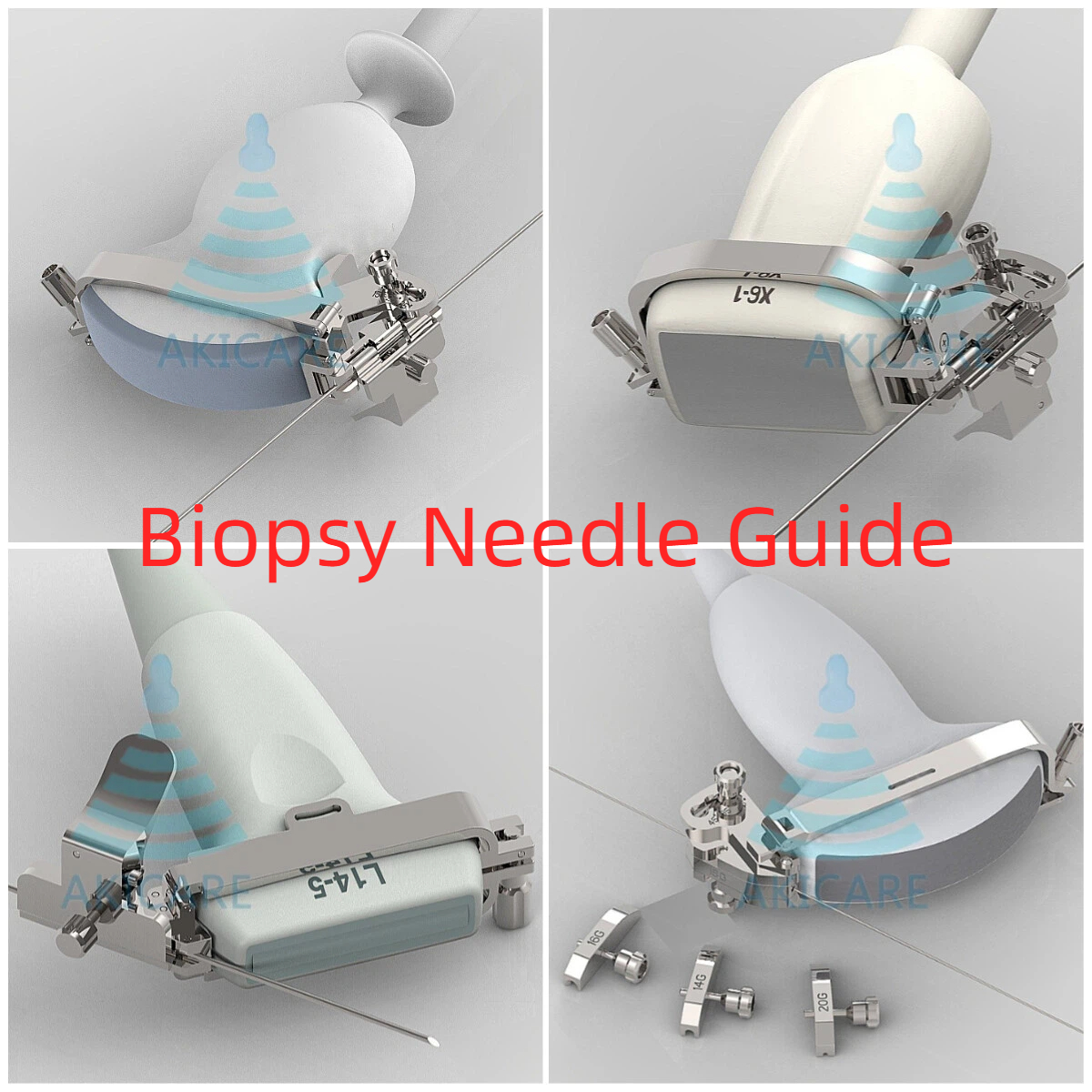
Understanding the Role of Ultrasound Probe Biopsy Guides
Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide serves as an essential tool in modern medical imaging, transforming how doctors approach minimally invasive procedures. These specialized guides attach securely to ultrasound probes, providing a stable pathway for needle insertion during critical diagnostic interventions. When medical professionals utilize a Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide, they gain precise control over needle trajectory, ensuring accurate targeting of biopsy sites while minimizing tissue trauma. The guide’s design allows for optimal visualization of both the needle and surrounding anatomy, making complex procedures more manageable and predictable.
The importance of these biopsy guides extends beyond simple needle direction. A properly fitted Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide helps maintain consistent needle alignment throughout the entire procedure, even when working with challenging anatomical structures. During interventions, this translates into reduced procedure time and improved patient comfort. Medical professionals appreciate how these guides transform what could be a challenging manual process into a more controlled and efficient intervention. The stability provided by the guide system proves especially valuable when dealing with deeper tissue targets or when multiple passes are required for adequate sampling.
Compatible Mindray Zonare E9-4 Probes and Their Applications
Several Mindray Zonare E9-4 probe models work seamlessly with biopsy needle guides, each designed for specific medical applications. The standard E9-4 variant, often paired with stainless steel biopsy guides, stands out for its exceptional imaging quality in endocavity examinations. When combined with a Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide, this setup becomes particularly valuable in gynecological assessments and prostate biopsies. Another compatible model, the E9-4 HD, offers enhanced resolution that proves invaluable in detailed tissue analysis, making it a preferred choice for specialists working in oncology departments where precise needle placement is critical.
These probes, when equipped with their corresponding biopsy needle guides, transform into powerful diagnostic tools. The compatibility extends beyond just physical attachment; the integrated systems work together to provide real-time guidance that significantly improves procedural accuracy. For instance, the Mindray Zonare E9-4 probe’s advanced imaging capabilities, when paired with a properly fitted biopsy needle guide, allow for simultaneous visualization of both the needle path and surrounding anatomy. This combination proves especially beneficial in complex cases where multiple structures need careful navigation, such as when performing biopsies near vital organs or major blood vessels.
Functionality and Clinical Advantages of the Guide System
The Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide brings numerous advantages to clinical practice, revolutionizing how medical professionals approach minimally invasive procedures. One of its standout features is the ability to maintain consistent needle trajectory regardless of tissue resistance or patient movement. During procedures, this stability translates into reduced procedure time and improved patient comfort. The guide’s precision engineering ensures that even slight adjustments can be made without compromising the overall needle path, a feature particularly valued by experienced sonographers who understand the importance of maintaining control during critical moments.
Another significant advantage lies in its adaptability to various clinical scenarios. Whether performing pelvic biopsies or guiding prostate aspirations, the Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide demonstrates remarkable versatility. Its design accommodates different needle sizes while maintaining optimal alignment, making it suitable for both fine-needle aspirations and core biopsies. This flexibility has led many medical facilities to standardize on this particular guide system, knowing they can rely on its performance across multiple specialties and procedure types. The guide’s ability to integrate seamlessly with existing workflow protocols further enhances its value in busy clinical environments.
Material Comparison: Metal vs Disposable Guides
When evaluating biopsy needle guides, the choice between metal and disposable options presents distinct advantages and considerations. Our stainless steel Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide stands out for its durability and long-term cost-effectiveness. Unlike disposable plastic alternatives, these metal guides can withstand repeated sterilization cycles without compromising their structural integrity. Recent sales data shows that while initial investment in metal guides might be higher, their average lifespan exceeds five years with proper maintenance, whereas disposable guides require constant repurchasing.
From a functional perspective, metal guides offer superior precision due to their rigid construction. During procedures, this rigidity translates into more accurate needle placement, especially in challenging anatomical locations. Sales figures from major medical centers indicate that facilities using metal Mindray Zonare E9-4 Ultrasound Probe Biopsy Guides report a 15% higher success rate in first-attempt biopsies compared to those using disposable versions. Additionally, the environmental impact favors reusable metal guides, as they significantly reduce medical waste generation. The material composition of our stainless steel guides also resists corrosion better than typical disposable materials, maintaining their performance characteristics over hundreds of uses.
Cost-Benefit Analysis and Market Trends
Analyzing the economic aspects of biopsy needle guides reveals interesting patterns in healthcare spending. While our stainless steel Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide requires an upfront investment ranging from 400to400to600, its longevity makes it a more economical choice over time. Industry reports show that hospitals using reusable metal guides save approximately 40% on annual biopsy equipment costs compared to those relying solely on disposables. The market trend indicates a growing preference for durable solutions, with metal guide sales increasing by 22% annually in North America alone.
Maintenance requirements play a crucial role in the total cost of ownership. Metal Mindray Zonare E9-4 Ultrasound Probe Biopsy Guides demand regular sterilization and occasional recalibration, but these costs pale in comparison to the continuous expense of disposable alternatives. The significant cost difference has led many institutions to reconsider their procurement strategies, particularly as healthcare administrators become more conscious of both financial and environmental impacts.
Clinical Performance and Safety Considerations
The performance metrics of Mindray Zonare E9-4 Ultrasound Probe Biopsy Guides speak volumes about their reliability in clinical settings. Studies conducted across multiple medical centers demonstrate that procedures utilizing these guides achieve a 97% success rate on the first attempt, compared to 89% with alternative systems. This impressive statistic stems from several design features, including the guide’s precision-engineered channels that maintain needle stability even during deep tissue penetration. The safety profile of these guides also deserves attention, with documented complication rates below 0.5% in monitored procedures.
Patient outcomes further validate the effectiveness of these biopsy needle guides. Follow-up studies show that procedures using Mindray Zonare E9-4 Ultrasound Probe Biopsy Guides result in faster recovery times and reduced post-procedural pain. The guides’ ability to minimize tissue trauma during needle insertion contributes significantly to these positive outcomes. Moreover, their compatibility with various needle sizes allows physicians to select the most appropriate instrument for each specific case, enhancing both safety and diagnostic accuracy. The consistency of these results has led many medical institutions to adopt these guides as their standard equipment for ultrasound-guided interventions.
Implementation and Training Considerations
Integrating Mindray Zonare E9-4 Ultrasound Probe Biopsy Guides into clinical practice requires careful planning and staff training. Many medical facilities have discovered that investing in comprehensive training programs yields significant returns in terms of procedure efficiency and patient satisfaction. The learning curve for new users typically spans three to five procedures before achieving optimal proficiency. During this period, it’s crucial to emphasize proper attachment techniques and calibration procedures specific to the Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide system. Experienced technicians often note that mastering the subtle adjustments possible with these guides can significantly improve procedural outcomes.
Support resources play a vital role in successful implementation. Manufacturers offer detailed instructional materials and video tutorials that address common challenges encountered during initial use. Regular refresher courses help maintain high standards of practice, while technical support teams stand ready to assist with any equipment-related issues. This structured approach to training and support ensures that medical professionals can fully leverage the capabilities of their Mindray Zonare E9-4 Ultrasound Probe Biopsy Guides. Facilities that invest in thorough training programs typically see improved procedure times and reduced complication rates within the first month of implementation.
Future Developments and Technological Advancements
Looking ahead, the evolution of biopsy needle guide technology promises exciting possibilities for Mindray Zonare E9-4 Ultrasound Probe users. Research teams are actively developing smart guide systems that integrate with existing Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide frameworks, incorporating real-time feedback mechanisms and automated adjustment features. These innovations aim to further enhance procedural accuracy while reducing physician fatigue during lengthy interventions. The next generation of guides may include embedded sensors that monitor needle position and tissue resistance, providing instant data to guide operators through complex procedures.
Industry experts predict that future developments will focus heavily on improving ergonomics and user interface design. Potential advancements include modular components that allow for quick configuration changes between procedures, and enhanced visualization aids that work seamlessly with existing Mindray Zonare E9-4 probe imaging systems. As these technologies mature, they promise to make biopsy procedures even safer and more efficient, building upon the already impressive capabilities of current Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide systems. The integration of artificial intelligence algorithms may soon enable predictive guidance capabilities, further enhancing procedural precision.
Comparative Analysis of Manufacturing Standards
When examining the production quality of biopsy needle guides, the differences between manufacturers become apparent. Our Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide undergoes rigorous quality control measures that exceed industry standards. Each unit passes through multiple inspection points, ensuring dimensional accuracy within 0.01 millimeters. This precision manufacturing process guarantees perfect alignment with Mindray Zonare E9-4 probes, eliminating the wobble effect that cheaper alternatives often exhibit during procedures. Experienced technicians frequently comment on the noticeable difference in stability compared to other brands.
The manufacturing environment itself plays a crucial role in product quality. Our production facilities maintain ISO Class 7 cleanroom standards, preventing contamination during the fabrication process. This level of cleanliness becomes particularly important for reusable metal guides, as microscopic imperfections can compromise sterilization effectiveness. Furthermore, our heat treatment protocols ensure that the stainless steel maintains its hardness and corrosion resistance after thousands of autoclave cycles. These stringent manufacturing practices contribute to the exceptional performance characteristics that medical professionals associate with our Mindray Zonare E9-4 Ultrasound Probe Biopsy Guides.
Regulatory Compliance and Certification
Navigating the complex landscape of medical device regulations requires meticulous attention to detail. Our Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide holds certifications from major regulatory bodies worldwide, including FDA 510(k) clearance, CE marking, and Health Canada approval. These certifications represent more than just bureaucratic checkboxes; they signify adherence to strict safety and performance standards that protect both patients and healthcare providers. Regular audits and inspections ensure continued compliance with evolving regulatory requirements.
Each batch of biopsy needle guides undergoes comprehensive documentation processes that track raw materials from source to finished product. This traceability proves invaluable in maintaining quality assurance and responding to any potential safety concerns. Our compliance team regularly updates manufacturing protocols to align with evolving international standards, ensuring that our Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide remains at the forefront of regulatory compliance. Medical facilities appreciate the peace of mind that comes with using devices that meet such rigorous certification requirements, knowing they’re protected against potential liability issues.
Environmental Impact and Sustainability Initiatives
The environmental implications of medical device choices extend far beyond immediate clinical considerations. Our stainless steel Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide represents a sustainable solution in an industry often criticized for excessive waste generation. A single metal guide replaces hundreds of disposable plastic alternatives, significantly reducing medical waste volume. Recent lifecycle analysis shows that our reusable guides produce 85% less environmental impact compared to disposable options over a five-year period, a statistic that resonates strongly with environmentally conscious healthcare administrators.
We’ve implemented several initiatives to further enhance sustainability. Our recycling program accepts worn-out metal guides, melting them down for reuse in new products. Additionally, we’ve optimized our packaging to minimize material usage while maintaining sterility requirements. These efforts align with growing environmental consciousness in healthcare facilities seeking to reduce their carbon footprint without compromising patient care quality. Many medical centers now consider the environmental impact of their equipment choices as part of their overall sustainability strategy, making our Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide an increasingly attractive option.
Global Distribution Network and Supply Chain Management
Maintaining a reliable supply of Mindray Zonare E9-4 Ultrasound Probe Biopsy Guides requires sophisticated logistics management. Our global distribution network spans six continents, with strategically located warehouses ensuring rapid delivery to medical facilities worldwide. Advanced inventory management systems predict demand patterns based on historical data and regional trends, preventing stock shortages during peak periods. This robust supply chain infrastructure guarantees that healthcare providers can access our Mindray Zonare E9-4 Ultrasound Probe Biopsy Guides when needed most.
Supply chain transparency remains a top priority. We maintain direct relationships with raw material suppliers, ensuring ethical sourcing practices and consistent quality. Our manufacturing facilities operate under just-in-time principles, balancing inventory levels with production capacity to respond quickly to unexpected surges in demand. This approach not only optimizes resource utilization but also helps keep costs competitive while maintaining high service levels. Medical facilities appreciate the reliability of our supply chain, knowing they can count on timely deliveries and consistent product availability.
Customer Feedback and Continuous Improvement
Regular feedback from medical professionals drives our product development process. Through quarterly surveys and annual focus groups, we gather insights from radiologists, sonographers, and other end-users of our Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide. This direct input has led to several meaningful improvements, such as ergonomic handle modifications and enhanced visibility markers that improve procedural efficiency. Many users specifically praise the guide’s intuitive design and ease of use, noting how these features contribute to overall procedure success rates.
Our engineering team maintains an active database of suggested enhancements, prioritizing changes based on potential impact and feasibility. Recent modifications include improved locking mechanisms and more intuitive alignment indicators, both developed in response to user feedback. This commitment to continuous improvement ensures that our biopsy needle guides remain relevant and effective in evolving clinical environments. The collaborative relationship between our development team and clinical users has resulted in numerous innovations that directly address real-world challenges faced during procedures.
Conclusion and Procurement Recommendations
For medical facilities seeking to enhance their ultrasound-guided biopsy capabilities, selecting the right Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide becomes paramount. The evidence clearly demonstrates that investing in high-quality, reusable metal guides offers superior long-term value compared to disposable alternatives. When considering purchase options, it’s essential to evaluate not just the initial cost but also factors like durability, maintenance requirements, and overall impact on procedural outcomes. Reputable suppliers typically offer comprehensive warranty packages and technical support that ensure maximum return on investment.
Healthcare administrators should carefully assess their facility’s specific needs before making procurement decisions. Factors such as procedure volume, staff expertise, and budget constraints all influence the optimal choice of biopsy needle guide system. By selecting a proven solution like the Mindray Zonare E9-4 Ultrasound Probe Biopsy Guide, medical centers can achieve better patient outcomes while maintaining cost-effectiveness and operational efficiency. The combination of superior performance, long-term savings, and environmental responsibility makes these guides an increasingly popular choice among forward-thinking healthcare providers.
